Advertisement
It is now estimated that 1 in 8 Americans are on serotonin reuptake inhibitors (SSRI) antidepressants1 and a shocking 1 in 4 among women in their 40s and 50s.2 Yet the U.S. suicide rate of 38,000 a year has never been higher.3
(Article by Dr. Mercola)
Clearly the glut of SSRI prescriptions is not lowering the national suicide rate; rather there is compelling evidence that the popular pills are actually contributing to suicide.
SSRIs and Violence
The first suspicion that SSRIs can cause dangerous and unintended psychiatric effects was a Kentucky shooting in 19894 in which pressman Joseph T. Wesbecker entered his former workplace, Standard Gravure, killed eight people, injured 12 and committed suicide after being prescribed Prozac.
Families of the wounded and killed soon filed a lawsuit against Prozac maker Eli Lilly and Company, claiming the SSRI contributed to the violence. The case went to a jury that sided with Lilly.
Yet three days before the shooting, Wesbecker’s psychiatrist had written “Prozac?” in his patient notes as a possible explanation of his bizarre behavior.
Since the Standard Gravure killings, psychiatrists, drug safety advocates and bereaved families have consistently tried to expose links between SSRIs and suicides but are hampered by mainstream safety data that deny a suicide link.
Study Suggests ‘No Suicide Link’ Is Not to Be Trusted
However, a recent study suggests the “no suicide links” findings are not necessarily to be trusted, noting that: “Therapists should be aware of the lack of proof from RCTs (randomized control trials) that antidepressants prevent suicides and suicide attempts.”5

Dr. David Healy, professor of psychiatry at Bangor University and author of 20 books including “The Antidepressant Era,” “The Creation of Psychopharmacology,” “Let Them Eat Prozac,” “Mania,” and “Pharmageddon,” heartily agrees that the SSRI statistics given to the public is problematic.6
“People haven’t had access to the data. There have been no publications around it. This is one of the biggest problems on which there’s a huge amount of data, but to which we’ve got little or no access … If we were getting our drug information from The New York Times instead of medical journals, we would all be a lot safer. When the Times reporter Jayson Blair was found to have fabricated stories, he was history. But the editors and writers involved with journal fraud still have their jobs and the articles are not even retracted. In fact, Liz Wager, Ph.D., the chair of the Committee on Publication Ethics (COPE) is herself Pharma-linked.”
The COPE website said about Wager,7 its former chair, “Liz provides writing, editing, training and consultancy services for various pharmaceutical companies” (most recently AstraZeneca, Cephalon, Cordis Corporation, GlaxoSmithKline, Eli Lilly, Janssen-Cilag, Merck Serono, Mundipharma, Norgine, Novo Nordisk, Sanofi Pasteur and Vifor Pharma) at the time of the interview with Healy.
Healy estimates as many as 1,000 to 2,000 Americans on SSRIs kill themselves each year, when they otherwise would not have done so. Violent acts against others and birth defects are also linked to the pills, he says.
Suicides Linked to Antidepressants Number in the Thousands
Even as high level links between medical editors and the drug industry prevented accurate information from reaching the public, in 1997, drug safety activists launched a website called SSRIstories.com,8 which archived credible and published reports that cite the role of SSRIs and related antidepressants in suicides and other violent behavior.
There are now thousands of entries. “The kind of energy, rage and insanity seen in a lot of crimes today was not seen before SSRIs appeared,” said Rosie Meysenburg, a founder of the website in an interview shortly before her death.9
In addition to the thousands of suicides, “there are two cases of women on the SSRI Stories site who stab a man close to 200 times and a case of a man who stabs his wife over 100 times and then goes next door to the neighbor’s house and stabbed the neighbor’s furniture about 500 times.”
The SSRI stories archive includes people on SSRIs setting themselves on fire, violent elderly people (which is rare) and bizarre cases of kleptomania and female school teachers molesting their minor male students. The common denominator in all the recorded crimes is the drug.
Drug companies routinely blame suicides on the depression that was being treated, not the drugs — but the experiences of patients treated with the same drugs for non-mental indications like pain and the experiences of healthy volunteers cannot be written off as the “disease.”
The Dark Side of Cymbalta
In 2004, 19-year-old Traci Johnson who had no history of mental problems hung herself in the Eli Lilly Clinic in Indianapolis while testing the drug giant’s serotonin–norepinephrine reuptake inhibitor (SNRI) duloxetine, sold under the brand name Cymbalta, a type of antidepressant similar to SSRIs.10
The suicide did not delay the drug’s approval and wide use. In 2008, the Journal of Clinical Psychopharmacology describes a 37-year-old man with a stable marriage, stable employment and no history of mental problems trying to kill himself two months after being prescribed Cymbalta for back pain.
“The patient was unable to state exactly why he wanted to commit suicide,” wrote the four physician authors in the report, also noting that the man returned to normal when the drug was stopped.
The authors also report a 63-year-old man with no mental health history becoming suicidal two weeks after being put on Cymbalta for fatigue, insomnia and sadness, yet he too was “unable to explain why he was having thoughts of wanting to die.”
Other cases of healthy people committing suicide on Cymbalta have been reported11and many still remember the suicide of Carol Gotbaum at Phoenix’s Sky Harbor International Airport who was on the drug. She was the stepdaughter-in-law of New York City’s public advocate at the time, Betsy Gotbaum.
Writing for Slate, reporter Jeanne Lenzer identified 13 suicides12 linked to Cymbalta besides Traci Johnson. Eli Lilly wanted to market the drug as a treatment for urinary incontinence too but withdrew its application and would not release the study data to Lenzer, she says. It may well have contained more evidence of suicide side effects.
The Drug Industry Still Fights Black Box Warnings Added in 2004
In 2004,13 in response to the outcry over antidepressant-linked suicides, the U.S. Food and Drug Administration (FDA) directed drug makers to add a “Black Box” warning to SRRIs and related psychiatric drugs, highlighting suicide risks and the need for close monitoring of children and adolescents for suicidal thoughts and behavior.
“Today’s actions represent FDA’s conclusions about the increased risk of suicidal thoughts and the necessary actions for physicians prescribing these antidepressant drugs and for the children and adolescents taking them. Our conclusions are based on the latest and best science. They reflect what we heard from our advisory committee last month, as well as what many members of the public have told us,” said Dr. Lester M. Crawford, acting FDA commissioner at the time.
Unfortunately, then and now, drug industry funded doctors have tried to claim that the warnings scare doctors and patients away and heighten suicide. While it would be ridiculous to blame obesity on tighter restriction of obesity drugs, that is essentially what drug industry spokesmen have done with SSRI warnings and continue to do.
Even The New York Times was misled by such disinformation, reporting that SSRI warnings were causing a leap in suicides.
Journalist Alison Bass, however, revealed14 the paper on which the Times article was based was funded by a $30,000 Pfizer grant. The conclusions about higher suicides also turned out to be wrong because the researcher got his years mixed up.15
Contrary to drug industry claims about the warnings, the proportion of children and teens taking antidepressants actually rose in the U.S. after the Black Box was added from more than 1 percent to nearly 2 percent says Dr. Andrea Cipriani, associate professor in the department of psychiatry at the University of Oxford, in England.16
Still, both David Shern, Ph.D., president of Mental Health America, a group investigated by Congress for undisclosed industry funding17 and Dr. Charles Nemeroff, also investigated by Congress, blamed18 the Black Box warnings for rising suicides. Speaking to ABC News, Nemeroff said:19
“I have no doubt that there is such a relationship. The concerns about antidepressant use in children and adolescents have paradoxically resulted in a reduction in their use, and this has contributed to increased suicide rates.”
False Charges About Black Box Warnings Continue
Nemeroff left his post at Emory University in disgrace because of his drug industry links20 and a National Institutes of Health (NIH) grant he managed was suspended because of the conflicts of interest — a rare occurrence.21 Nor have the false charges about Black Boxes died down. Here is how a New York Times editorial read just last year.22
“Worse, antidepressants, which can be lifesaving, are probably being underused in young people. Their use fell significantly after the FDA issued its so-called black-box warning in 2004, stating that all antidepressants were associated with a risk of increased suicidal feeling, thinking and behavior in adolescents. That warning was later extended to young adults. It’s not hard to understand why. The FDA’s well-intended warning was alarming to the public and most likely discouraged many patients from taking antidepressants. Physicians, too, were anxious about the admittedly small possible risks posed by antidepressants and were probably more reluctant to prescribe them. This very small risk of suicidal behavior posed by antidepressant treatment has always been dwarfed by the deadly risk of untreated depression … Parents and teenagers, and their doctors, too, should not be afraid of antidepressants and should know that they can be very helpful. Indeed, with careful use and monitoring, they can be lifesaving. The only thing we should all fear is depression, a natural killer that we can effectively treat.”
Blaming underuse of drugs and falling sales on warnings that made patients or doctors “anxious” is not limited to antidepressants. Recently, industry-funded groups charged that warnings on the bone drugs called bisphosphonates about fractures and osteonecrosis of the jaw were scaring patients and doctors away and denying patients the drugs’ benefits.23
SSRIs Ignored in the Extremely High Rate of Suicide in the Military
During the wars in Iraq and Afghanistan, troop suicides were higher than combat fatalities themselves and the majority of the suicides were among troops who had never even deployed.24 But when a long awaited Army report came out, it largely blamed soldiers themselves for the deaths, especially highlighting illegal drug usage and barely mentioning the huge number of troops on prescription psychoactive drugs. In fact, the word “illicit” appears 150 times in the Army report and “psychiatrist” appears twice.25
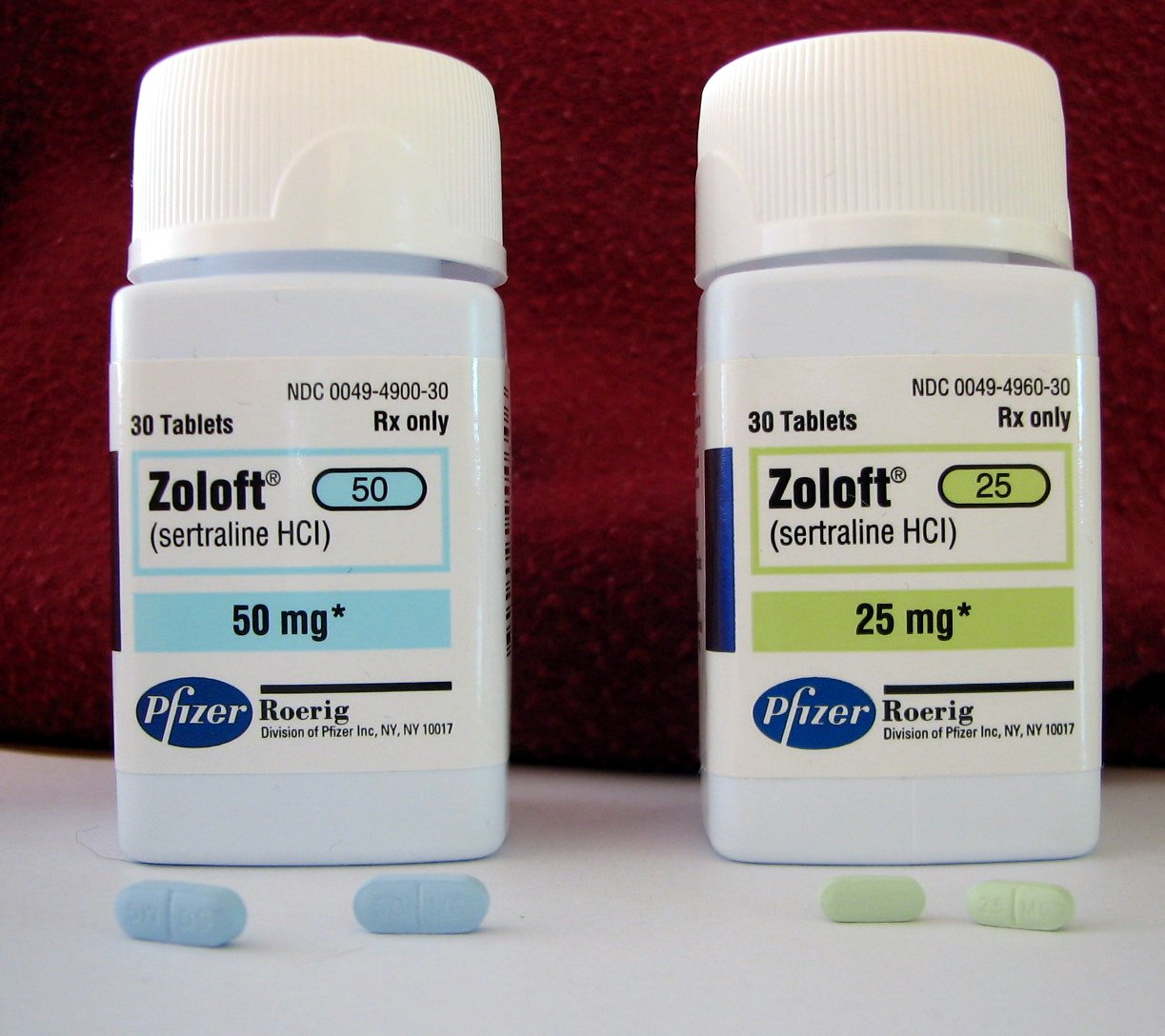
At the time of the Army report, 73,103 prescriptions for Zoloft had been dispensed to troops, 38,199 for Prozac, 17,830 for Paxil and 12,047 for Cymbalta.26 In fact 4,994 troops at Fort Bragg alone were reported to be on antidepressants by the Fayetteville Observer.
Four years after the Army report, researchers addressed the military suicides in JAMA Psychiatry27 again not finding or considering the high prescribing of SSRIs within the military. The authors had financial links to Eli Lilly, GlaxoSmithKline, Ortho-McNeil Pharmaceutical, Janssen-Cilag, Pfizer, Sanofi-Aventis, Shire and Johnson & Johnson.
In a series during the Iraq and Afghanistan wars called “Medicating the Military,” when SSRI use was mushrooming, Military Times reported:28
“A Military Times investigation of electronic records obtained from the Defense Logistics Agency shows DLA spent $1.1 billion on common psychiatric and pain medications from 2001 to 2009. It also shows that use of psychiatric medications has increased dramatically — about 76 percent overall, with some drug types more than doubling — since the start of the current wars. Troops and military health care providers also told Military Times that these medications are being prescribed, consumed, shared and traded in combat zones — despite some restrictions on the deployment of troops using those drugs. The investigation also shows that drugs originally developed to treat bipolar disorder and schizophrenia are now commonly used to treat symptoms of post-traumatic stress disorder, such as headaches, nightmares, nervousness and fits of anger. Such ‘off-label’ use — prescribing medications to treat conditions for which the drugs were not formally approved by the FDA — is legal and even common. But experts say the lack of proof that these treatments work for other purposes, without fully understanding side effects, raises serious concerns about whether the treatments are safe and effective.”
Many military administrators have unabashed drug company links, like Dr. Matthew Friedman, former executive director of the Veterans Affairs’ National Center for PTSD,29 who admitted receiving AstraZeneca money in a video on the Center’s site a few years ago (a video since taken down) and served as Pfizer Visiting Professor while helming a government organization.30
Recently, the Annals of Internal Medicine ran another study looking at military suicides without finding an antidepressant role. The study’s editors at the Annals had links31 to Eli Lilly, Pfizer and Johnson & Johnson. Considering all the risks associated with antidepressants, it would be wise to use them as a very last resort. To learn more about safer treatment options, please see my previous article, “Supplements Proven Beneficial for Your Mental Health.”
Read more at: articles.mercola.com
Submit a correction >>
Advertisement
Advertisements