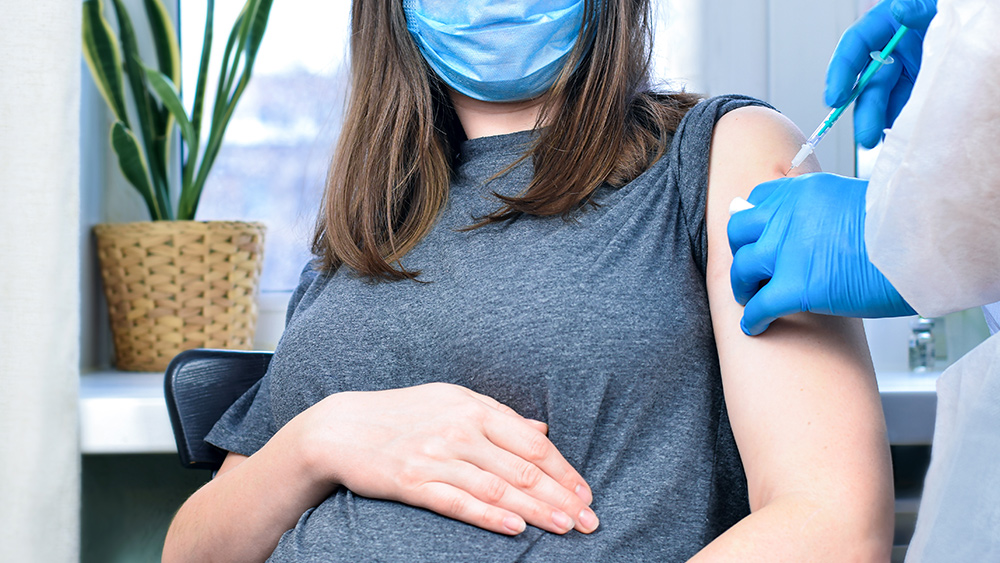
The American College of Obstetricians and Gynecologists (ACOG) received $11.8 million from the Department of Health and Human Services (HHS) to encourage pregnant women to get the Wuhan coronavirus (COVID-19) vaccine despite the fact that they were excluded from clinical trials and vaccine safety had not been tested.
ACOG is the largest and most respected organization of obstetricians and gynecologists in the United States. According to the Freedom of Information Act (FOIA) request filed in 2022 by healthcare advocate Maggie Thorp, ACOG received three grants from the HHS and the Centers for Disease Control and Prevention (CDC) during the pandemic. (Related: Largest organization of OB-GYNs in America accepted $11 million from HHS to promote COVID-19 vaccines to PREGNANT WOMEN.)
On February 1, 2021, ACOG was awarded a cooperative agreement grant by HHS and the CDC for the first time. The contract stated that ACOG had to cede over projects funded by the CDC and align with CDC guidance on COVID-19.
Moreover, if ACOG fails to properly promote the safety and effectiveness of COVID-19 vaccines for pregnant women and new mothers, ACOG will be forced to return all of the money it received to the HHS.
As part of the compliance, ACOG joined the HHS in its "COVID-19 Community Corps" program on April 1, 2021, to create a network of local community leaders who could encourage pregnant women and new mothers to get vaccinated against COVID-19.
The HHS and the CDC provided information about public health and facts about COVID-19, while Vice President Kamala Harris and Surgeon General Vivek Murthy met with the founding members to plan a public education campaign from the White House promoting vaccinations and building confidence in the vaccines.
Furthermore, when former CDC Director Dr. Rochelle Walensky started recommending all pregnant women to get vaccinated on April 23, 2021, former ACOG president Dr. J. Martin Tucker along with Dr. William Grobman, the president of the Society of Maternal-Fetal Medicine, also started doing so two months later despite the absence of clinical trial data.
ACOG also pushing pregnant women to get vaccine boosters
In addition to recommending COVID-19 vaccines without substantial data, ACOG expanded its recommendations to include new bivalent COVID-19 booster vaccines, despite a lack of approval from the U.S. Food and Drug Administration (FDA).
"Vaccination may occur in any trimester, and emphasis should be on vaccine receipt as soon as possible to maximize maternal and fetal health. This recommendation applies to both primary series and booster vaccination," ACOG stated on their website.
Additionally, ACOG stated that COVID-19 vaccines could be administered with other vaccines, such as influenza and Tdap vaccines at the same time, despite the absence of clinical trials demonstrating the safety of co-administering multiple vaccines to pregnant women.
However, the FDA's healthcare provider fact sheets for the Moderna and Pfizer bivalent vaccines both stated otherwise:
"Available data on Moderna COVID-19 vaccine administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy. Data are not available on Moderna COVID-19 vaccine, Bivalent, administered to pregnant women."
"No data are available regarding the use of Pfizer-BioNTech COVID-19 Vaccine, Bivalent during pregnancy. Available data on Pfizer-BioNTech COVID-19 Vaccine administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy."
Meaning to say, both noted the absence of data on the use of Moderna and Pfizer vaccines during pregnancy, emphasizing the insufficient information regarding vaccine-associated risks. However, despite these uncertainties, the recommendation of ACOG remained firm.
Watch Ben Armstrong discussing the government's bribery of ACOG to push the COVID-19 vaccines on "The Ben Armstrong Show."
This video is from the channel The New American on Brighteon.com.
More related stories:
If your doctor told you to get "vaccinated" for COVID, they were BRIBED, evidence shows.
Pfizer data reveals COVID-19 vaccines were NEVER safe for pregnant women.
DATA ANALYSIS: Increase in miscarriages and stillbirths directly linked to COVID-19 vaccines.
Sources include:
Please contact us for more information.