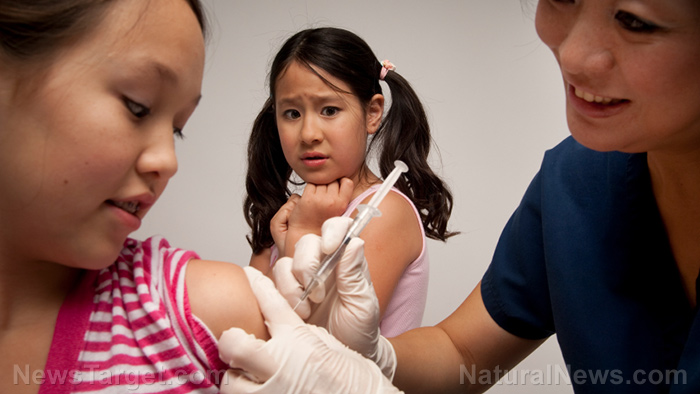
After developing coronavirus (COVID-19) vaccines for adults, Big Pharma companies are now aiming for a younger demographic.
Vaccine manufacturers have commenced trials for Wuhan coronavirus jabs on children. However, the need to immunize children against the disease raised serious questions as juveniles have a very high COVID-19 survival rate. Furthermore, children have very low chances of being hospitalized and dying from it.
Four vaccine makers have started conducting clinical trials of their respective COVID-19 jabs to scrutinize the effects of the shots on younger patients.
In January, Moderna announced that it began vaccine trials for young volunteers – enrolling 3,000 volunteers between 12 and 17 years old. The Boston-based firm will use the same vaccine doses for adults for the new clinical trials. Researchers from the University of Massachusetts Medical School will be in charge of the vaccine trials.
Meanwhile, Pfizer also expanded its clinical trials to include children aged 12 to 15 years old. CNBC reported in the same month that the firm's vaccine trials for younger volunteers were already fully enrolled. Its vaccine, developed with German company BioNTech, received emergency approval for use with people 16 years old and above.
In February, British drug manufacturer AstraZeneca began testing its vaccine on children. The University of Oxford, AstraZeneca's partner, said its clinical trials that month will look at the immune response on volunteers aged six to 17 years old triggered by the vaccine.
Pediatric infection and immunity professor Andrew Pollard said: "While most children are relatively unaffected by the [Wuhan] coronavirus, … it is important to establish the safety and immune response to the vaccine in children and young people.
Johnson & Johnson also announced on Feb. 28 that it intends to test its one-dose Wuhan coronavirus jab on infants, pregnant women and people with compromised immune systems. The Baltimore Sun reported that a spokesperson for Janssen Biotech – the J&J subsidiary in charge of the jab – confirmed the trials.
Dr. Ofer Levy, a member of the Food and Drug Administration (FDA) advisory committee that examined J&J's vaccine data, remarked: "[J&J] did not get into a lot of detail about it, but [the company] did make it clear [it] will be pursuing pediatric and maternal coronavirus immunization studies." Levy personally believes that children need not get the jab as a requirement for schools to reopen.
The necessity of a Wuhan coronavirus vaccine for kids comes into question due to their very high survival rate
There are huge questions regarding the need to vaccinate children for COVID-19. Earlier studies have shown that the survival rate for birth-age to 19-year-old individuals is 99.997 percent. A 2021 paper noted that hospitalizations and hospital deaths for children diagnosed with COVID-19 are rare – with only 0.19 percent dying from the disease.
Furthermore, the vaccines are primarily designed to address the symptoms experienced when someone contracts the Wuhan coronavirus. It does not prevent them from transmitting the pathogen responsible for COVID-19. Even the World Health Organization (WHO) remarked that there is no guarantee that the COVID-19 jabs will prevent people from being infected with SARS-CoV-2 pathogen and transmitting it to others. (Related: Coronavirus transmission rates: Every ONE patient infects TWO to THREE other people.)
During a Dec. 28 virtual press conference, WHO Chief Scientist Soumya Swaminathan said the global health body currently has no evidence that any of the vaccines could "prevent people from actually getting the infection and … being able to pass [the virus] on." She added: "We need to assume that people who have been vaccinated also need to take the same precautions [as those who haven't.]"
WHO Health Emergencies Program Executive Director Dr. Mike Ryan seconded Swaminathan's remarks. He commented: "The existence of a vaccine, even at high efficacy, is no guarantee of eliminating or eradicating an infectious disease."
National Institute for Allergy and Infectious Diseases Director Dr. Anthony Fauci echoed the WHO's opinion on vaccines during a Jan. 1 Newsweek interview. He said health officials do not know if COVID-19 vaccines prevent infection, or if people who got inoculated can still spread the Wuhan coronavirus to others. "We do not know if the vaccines that prevent clinical disease also prevent infection. They very well might, but we have not proven that yet," the country's top infectious disease doctor told the magazine. (Related: WHO backtracks on coronavirus vaccines, admits they won't work.)
In December 2020, the FDA authorized the Pfizer/BioNTech BNT162b2 and Moderna mRNA-1273 vaccines for emergency use in the U.S. However, the companies only presented clinical trial data showing that their jabs prevented mild to severe COVID-19 symptoms in vaccinated volunteers. They did not examine if their vaccines prevented people from contracting asymptomatic coronavirus infection and transmitting the pathogen to others.
Visit Vaccines.news to read more about vaccines for addressing the Wuhan coronavirus pandemic.
Sources include:
Please contact us for more information.