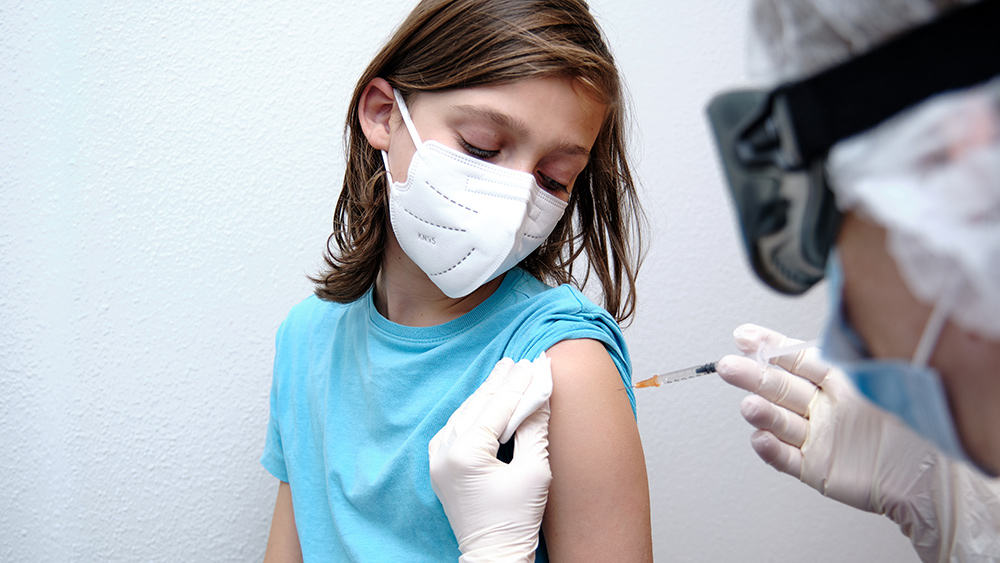
Hartford Healthcare in Connecticut started giving kids the Pfizer Wuhan coronavirus (COVID-19) vaccine just minutes after the Centers for Disease Control and Prevention (CDC) officially approved its use for children aged five to 11.
The hospital reportedly vaccinated six children with low doses of Pfizer's vaccine, causing a storm of controversy as there is zero evidence that vaccinating children is necessary. Data has shown that COVID poses little to no risk to the age group, with children accounting for less than 0.1 percent of COVID deaths in America.
On Oct. 29, the Food and Drug Administration (FDA) approved the emergency use authorization of Pfizer's COVID-19 vaccine for children aged five to 11. The decision puts the U.S. as one of the first countries in the world to officially approve a COVID-19 vaccine from that age group.
Meanwhile, British scientists warned officials against "blindly" following their footsteps as far as vaccination is concerned, advising them to weigh the risks "extremely carefully."
David Livermore, a medical microbiologist at the University of East Anglia said that vaccinating children to protect adults via herd immunity is "ethically dubious and scientifically weak." With fears of myocarditis detected in children, many critics say that they are better off catching COVID and getting natural protection than giving them injections with known adverse effects.
Adverse effects of vaccine on kids more concerning than COVID
The CDC convened a panel of independent scientists to review the available data on the status of COVID-19 outbreak in children and the effectiveness of Pfizer's vaccine as well as its possible side effects. The panel unanimously recommended the vaccine, with the CDC endorsing the recommendation.
However, the main concern was the risk of myocarditis, which is an inflammation of the heart muscle detected in mostly male adolescents and young adults after vaccination with Pfizer or Moderna shots.
There are also signs that natural immunity is already slowing down the epidemic.
Most cases of myocarditis after a COVID injection are mild and treatable, but the U.K. government's scientific advisers say the long-term effects of the inflammation are not yet understood. (Related: Study: 397 Children diagnosed with heart inflammation after receiving Pfizer's COVID-19 vaccine.)
In what can be depicted as child abuse, the so-called data used to justify the FDA panel's earlier decision showed that nearly 180 children would be expected to suffer from myocarditis for every death the vaccine would prevent if the rollout continues.
The Pfizer dose for children is only one-third of the original dose for adults and is given in two doses, three weeks apart. This lower dose was chosen to minimize side effects while continuing to produce strong immunity. The caps on children's vials will also be orange, to make them recognizable from the purple caps on the vials for older groups.
Dr. Michael Kurilla, the director of the Division of Clinical Innovation at the National Institutes of Health (NIH) National Center for Advancing Translational Sciences, noted that while there may be children at high risk of severe COVID due to underlying conditions who could benefit from the vaccine, he is not sure if this is applicable to all kids in the five to 11 age group.
He also mentioned that those who have been infected with COVID in the past already have immunity. He added that current data does not suggest that the protection will last long enough for children and that the antibodies from the vaccine will eventually wane.
Meanwhile, the government has already secured enough vaccines for every child in the U.S., with the White House saying that vaccination will be fully up and running during the week of November 8.
Read more about how the COVID-19 vaccines can affect children at Immunization.news.
Sources include:
Please contact us for more information.